
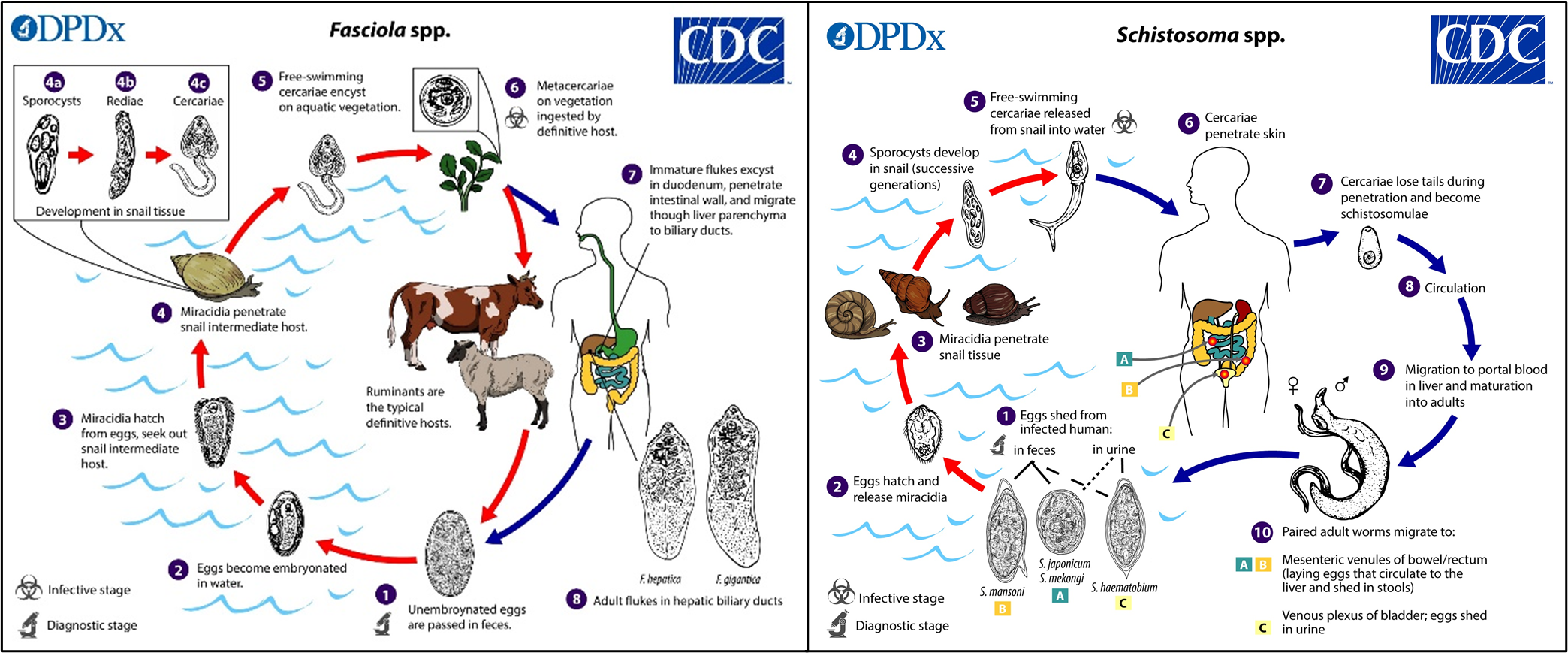
Trematodes
Trematodes, or parasitic flatworms/flukes, are a large and highly diverse group of globally distributed parasites that can be broadly characterised as blood flukes (e.g., Schistosoma species), liver flukes (e.g., Fasciola, Chlonorchis and Ophistorchis species), lung flukes (e.g., Paragonimus species), and intestinal flukes (e.g., Metagonimus species), depending on where their adult form live within the host.
Trematodes from all four groups cause untold suffering to hundreds of millions of humans and animals, resulting in significant morbidity, mortality, and socioeconomic loss worldwide. Of particular note are the schistosome blood flukes and fasciolid liver flukes, causative agents of schistosomiasis and fascioliasis considered Neglected Tropical Diseases (NTDs) by the WHO. The most prevalent and widely distributed species are Schistosoma mansoni and Fasciola hepatica.
Treating flatworm infections
Praziquantel (PZQ) has been used worldwide for over 40 years to treat human and animal flatworm infections, as well as some tapeworms.
Only recently has its mechanism of action been uncovered. PZQ binds to TRPMPZQ (a transient receptor potential ion channel in the melastatin family) via a binding pocket within the receptors voltage-sensor like domain (VSLD), causing an influx of calcium ions into the parasite resulting in muscle contraction, rapid paralysis, damage to the parasite’s tegument, and death.
Although not without its (considerable) downsides, PZQ has proven highly effective in treating many parasitic worm infections, and is the main control strategy used against these parasites. PZQ is not, however, effective against Fasciola species due to natural amino acid variation exclusive to fasciolids that renders PZQ unable to activate the TRPM channel. As such, an alternative drug, triclabendazole (TCBZ), has been used widely to treat human and animal fascioliasis.
Worryingly for global food security and public health, resistance to TCBZ has been detected in fasciolids on every continent, and the risk of PZQ resistance evolving is of great concern. Given the risk to control successes posed by TCBZ resistance, and limitations and concerns with PZQ, the WHO have highlighted a crucial need for alternative to PZQ.
Developing a novel broad-spectrum flukicide
In a recent and incredibly exciting study, Daniel Sprague and colleagues from the Medical College of Wisconsin, Scripps Research (USA), and Justus Liebig University Giessen (Germany) report on the development of a highly promising broad-spectrum flukicide.
Building on a 2021 study from the same group which used high-throughput target-based screening of ~16,000 compounds to find activators of the S. mansoni TRPMPZQ (Sm.TRPMPZQ) receptor, a novel N-benzamidoquinazolinone (‘compound 1’) was identified that dual-activated both Sm.TRPMPZQ and Fh.TRPMPZQ.
In an attempt to optimise and improve the activity of ‘compound 1’, the sterics, or spatial arrangement of atoms in the core of the compound, as well as the northern and southern hemispheres, were modified and the activity of the modified compounds against Sm.TRPMPZQ and Fh.TRPMPZQ were analysed using a structure-activity analysis.
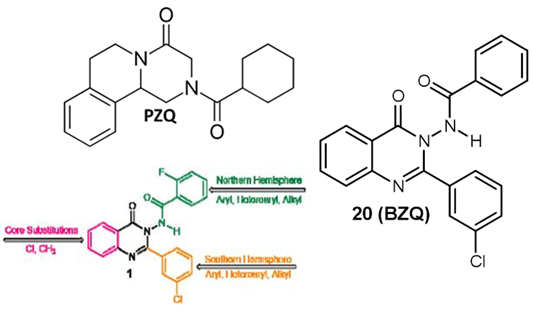
Overall, 42 ‘compound 1’ modifications were tested, many of which showed complete inactivity against TRPMPZQ receptors from both species, or reduced activity compared to ‘compound 1’.
One modified compound, however (‘compound 20’, referred to as BZQ (N–benzamidoquinazolinone)), promisingly showed improved potency against both Sm.TRPMPZQ and Fh.TRPMPZQ as well as against other fluke TRPMPZQ channels, including S. haematobium, S. japonicum, F. gigantica, Echinostoma caproni, Clonorchis sinensis, and Opistorchis viverrini. BZQ thus demonstrates potent, broad-spectrum TRPMPZQ channel activity against every fluke profiled in their study.
As PZQ activation of Sm.TRPMPZQ results in calcium ion influx (changing cellular membrane potential), electrophysiological analyses were conducted to compare the activity of BZQ and PZQ against Sm.TRPMPZQ, and BZQ against Fh.TRPMPZQ. These analyses supported BZQ as a potent activator of both Sm.TRPMPZQ and Fh.TRPMPZQ, and BZQ showed similar electrophysiological properties to PZQ activation of Sm.TRPMPZQ.
How does BZQ fair against live worms, and match up to PZQ and TCBZ?
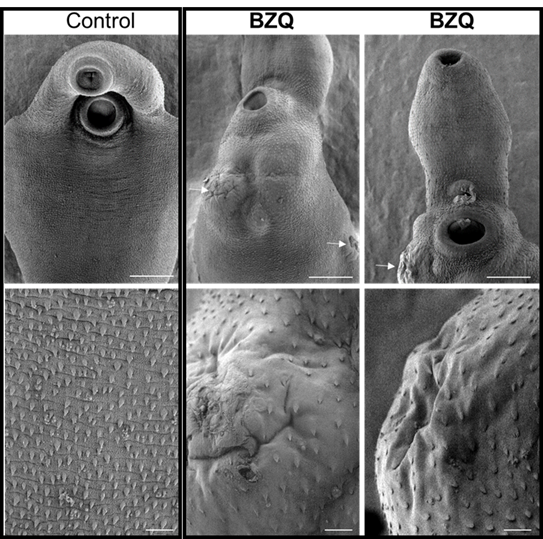
Next, the effect of PZQ or BZQ on live S. mansoni and F. hepatica adult worms was evaluated ex vivo, which showed that BZQ caused rapid paralytic muscle contraction in both worms, and confirmed PZQ only caused these contractions in S. mansoni. The impact on parasite surface tegument ultrastructures were subsequently assessed using electron microscopy, where BZQ-treated worms displayed considerable damage to their tegument similar to that seen in PZQ-treated S. mansoni worms.
BZQ was also demonstrated to be considerably more effective than TCBZ against adult and immature F. hepatica, where BZQ substantially reduced worm motility at lower concentrations and at a much faster rate than TCBZ.
In S. mansoni infected mice, BZQ was validated as a highly promising candidate flukicide, where BZQ-treated mice saw a 70% reduction in worm burden, compared to a 68% reduction using the currently employed PZQ.
Outcomes, impact, and perspective
This research discovered a novel compound (BZQ) using target-based screening and structure-activity analyses, and validated the activity, binding, and interaction of BZQ with the TRPMPZQ channels of the globally important parasitic flatworms S. mansoni and F. hepatica.
BZQ demonstrated activity against both Sm.TRPMPZQ and Fh.TRPMPZQ, and appears as effective against S. mansoni as PZQ, and more effective against F. hepatica than TCBZ. As well as these two flukes, BZQ also displayed activity at all other tested fluke TRPMPZQ channels, highlighting its broad-spectrum efficacy.
BZQ is therefore a highly promising flukicide with the potential to overcome limitations associated with PZQ and TCBZ, bolstering our arsenal of anthelmintic drugs and improving our ability to fight these devastating parasitic diseases.

Comments