
As a genomics journal, genome sequencing is our bread and butter, and we have had a couple of large-scale genome sequencing studies in crops. Lin Zeng and colleagues looked at pistachio. They sequenced the genomes of 93 different pistachio cultivars, and 49 wild trees, including some from different species of Pistacia. By comparing the genomes, they were able to date the domestication of the species to around 8000 years ago, and find that, perhaps unsurprisingly, farmers have selected trees with larger seed size.
A similar study was performed by Jinyu Wang and colleagues to compare the domestication of soybean and maize. For each crop, the authors compared the genomes of wild and cultivated plants. Interestingly, domestication has lead to similar changes in the genomes of both crops, with some signals being found preferentially in regions of the genome with no genes in. Both crops also show evidence of mutations caused by exposure to solar ultraviolet light.
It is not unusual for the molecular detail of traits to be less well explored in plants than in mammalian cells, but increasingly studies are changing that.
In recent years, there has been a lot of excitement about the potential of the CRISPR/Cas9 system for manipulating genomes. The system allows researchers to make cuts at specific places in the genome. After making the cuts, what happens next depends on the aims of the researchers. The cut can be used to inactivate a gene, or it can be repaired using an alternative sequence to change the behavior of the gene. A couple of recent studies we’ve published has used CRISPR/Cas9 in plants in interesting ways.
Devang Mehta and colleagues were trying to use CRISPR/Cas9 as a way of making cassava plants resistant to cassava mosaic geminivirus, a devastating disease that can seriously affect production of a crop consumed by over a billion people throughout Africa and south Asia. They engineered cassava plants to express CRISPR/Cas9 designed to cut sequences inside the virus genome, preventing the virus from reproducing in the plants. This is very similar to the role of CRISPR/Cas9 in its endogenous context in bacterial genomes, and has been used to engineer virus resistance in other crops. In this case, though, the authors were not successful, finding that the virus rapidly evolved to evade the cut. This provides food for thought for future efforts in this direction.
Haroon Butt and colleagues have used CRISPR/Cas9 as the basis of a method for mutating specific proteins in plants, with the potential of improving crops. As a proof of principle, they use it to derive rice resistant to splicing inhibitors targeting a specific splicing protein, SF3B1. Naturally occurring inhibitors of this protein are produced by species of Streptomyces bacteria, and other inhibitors have been used as herbicides. In the method, CRISPR/Cas9 is used to cut at all sites in the SF3B1 gene, and the cuts are repaired, potentially creating mutations. Plants are then grown in the presence of SF3B1 inhibitors, and those that grow are obviously resistant to the inhibitor. Different plants had different levels of resistance. The method could be applied to any gene known to respond to a given selection pressure, so could potentially be used to generate plants resistant to drought, or infectious disease, or high salt conditions, or any other condition that affects plant growth.
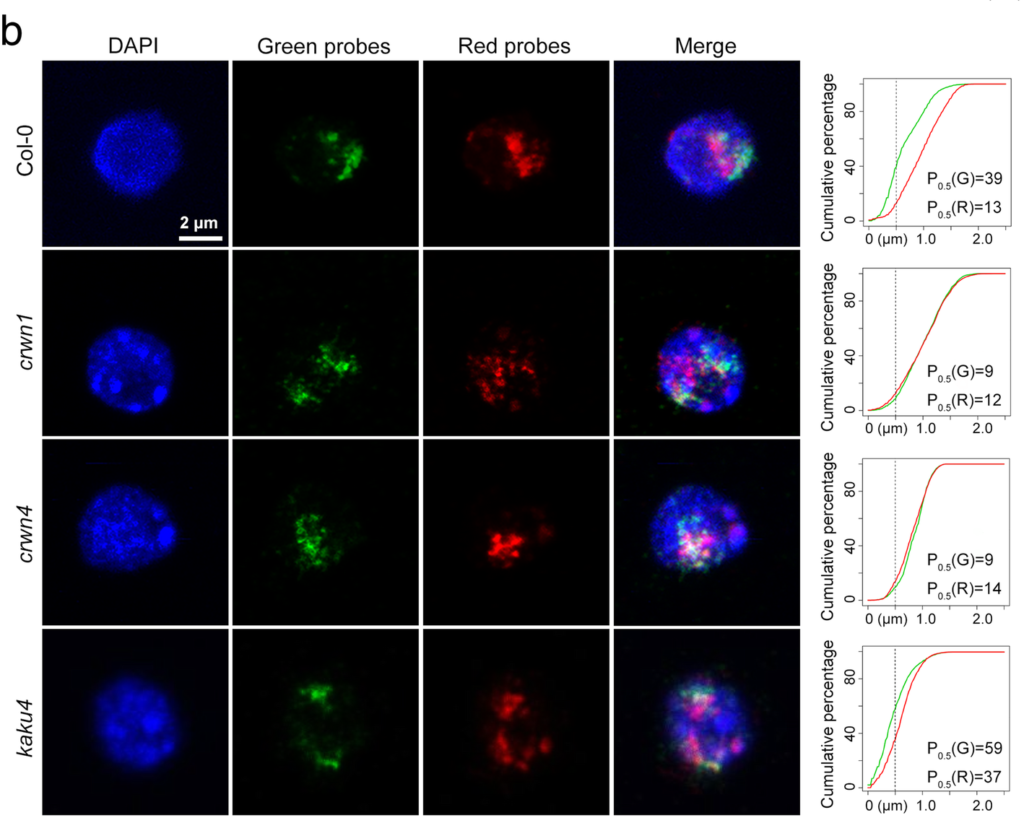
Finally, we have a nice molecular biology study. In mammals, it is well known that the nuclear lamins, proteins which form a basket-like structure to give the nucleus a shape, tether chromosomes to the periphery of the nucleus. The tethered sequences are enriched for transcriptionally silenced genes. In plants, it is also known that transcriptionally silenced genes localize to the nuclear periphery, but the details of this are much less well explored than in mammalian cells. Bo Hu and colleagues have looked at this in more detail. They show that, as in mammals, lamin-like proteins are required for positioning chromosomes at the edge of the nucleus, and that the DNA needs to be methylated at specific sequences for this to occur. One of the lamin-like proteins directly interacts with the DNA.
Sadly, despite plants being so fascinating, it is not unusual for the molecular detail of traits to be less well explored in plants than in mammalian cells, but increasingly, studies like the Hu one are changing that. We are also increasingly seeing innovative approaches to crop improvements, like those from Mehta and Butt, and more and more studies of the domestication history of crops, such as the Zeng and Wang articles described here. Plant genomics research really is flowering.
Andrew Cosgrove
Latest posts by Andrew Cosgrove (see all)
- Exploring the history of smallpox vaccination with 19th Century American vaccination kits - 16th November 2020
- The fascination of plant genomes - 20th May 2019
- Genome Biology at Genome Informatics - 18th February 2019
Comments